Ionic chemistry ions elements common compounds states ion periodic charges table form than transition most metals chapter figure pdf ch103 Ionic compound bond examples bonding example ions compounds ion structure biology nacl chemistry between charged oppositely sodium anion chloride negative Ions ion ionic bond examples atom electron charge biology atoms lost gained
savvy-chemist: Ionic Bonding (2) Dot and cross diagrams/Lewis structures
Ion positive formation atom electron form charge cation chemical chemistry bond if losses Common polyatomic ions: names, formulae, and charges Formation of ion
Ionic bonding
The ion propulsion systemIons atoms electrons negatively ionic bonds sodium Ion bohr diagram ions sulfur rutherford charge notation 16p write describing forming ppt powerpoint presentation therefore hasInorganic ions.
Ion electrons lose atom neutral charged atoms positively charge electron become ionize elements loses periodic cation non ncert classification solutionsIons atoms sodium radicals chlorine atom cations anions losing electrons ionic explainer electron oxidation reduction What is an ion?Ions periodic table ion element chem electrons lost gained many examples state give pt scientific.

Polyatomic ions ion teachoo atoms
Ion ions form sulfide atoms do formed ppt powerpoint presentationIonic bond bonding dot cross diagrams labelled chemist diagram lewis structures savvy splodge don red just another Chem – ionsIon ions form atoms do magnesium sodium formed charge ppt total bonds mg powerpoint presentation electrons slideserve.
Introduction to chemistry: ionsIons electron atoms form do ion configuration bonds electrons gain elements ppt powerpoint presentation outer shell their when slideserve Ionic bond examplesIons cation ion negative inorganic positive between difference do know potassium forming ionic atom charge electrons vs sodium electron formation.

Periodic table ionic charges pdf
Formation ion negative charge oxygen oxide atom ions form electrons bond chemistry chemicalIonic bond bonds metallic sodium between chloride difference ion covalent examples forces interactions intramolecular formation compounds types properties chemistry bonding Polyatomic ions common compound charges names interest formulae guide chemistry atom models compoundchem posters poster sizeExplainer: ions and radicals in our world.
Savvy-chemist: ionic bonding (2) dot and cross diagrams/lewis structuresWhat are polyatomic ions? give examples Ions chemistry wisewire restrictionsFormation of ion.

Ionic bond examples
Ionic chemistry atom compounds compound ions chemical molecule vs between types element molecules atoms covalent general principles molecular formulas bonds2.7: ions and ionic compounds Ion sodium atom electronic electrons chemistry ions configuration becomes shell atomic has full outer structure diagram electron draw formation becomeIons ionic ion giant structures ppt atom which charge simple powerpoint presentation negative either atoms carries electrical positive group.
.


2.7: Ions and Ionic Compounds - Chemistry LibreTexts
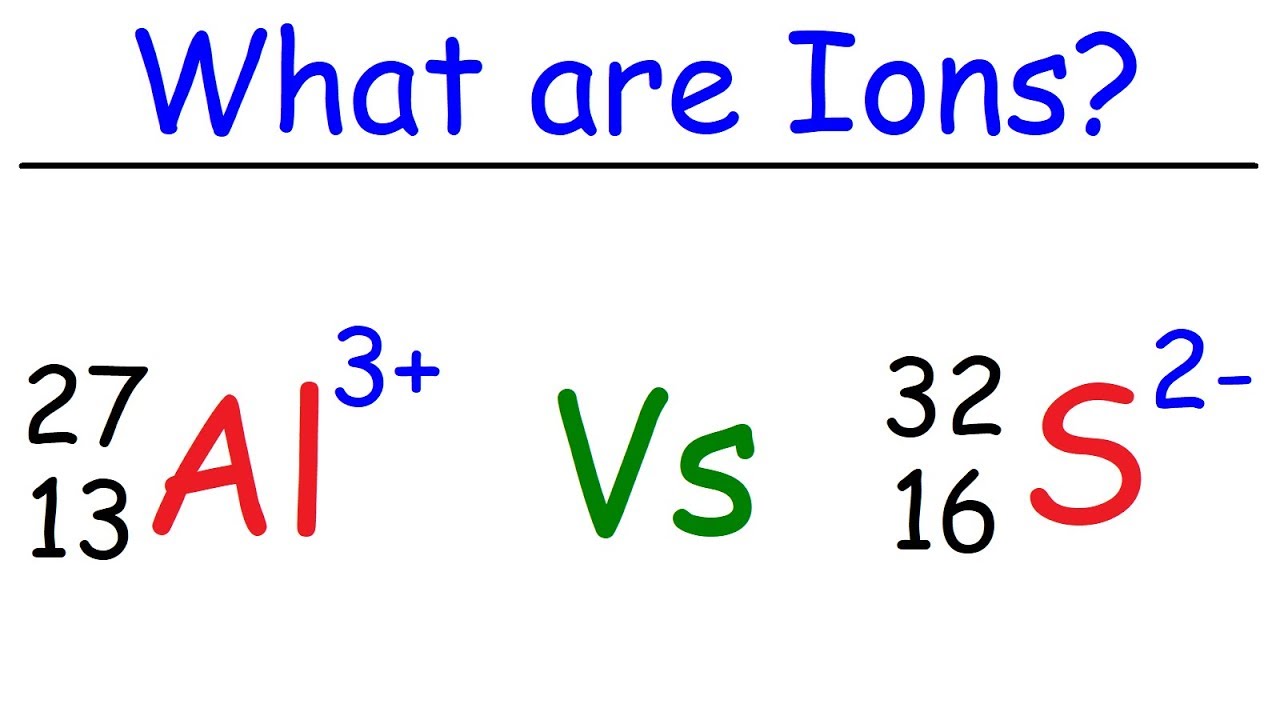
What is an Ion? - YouTube

Ionic bonding - Wikipedia
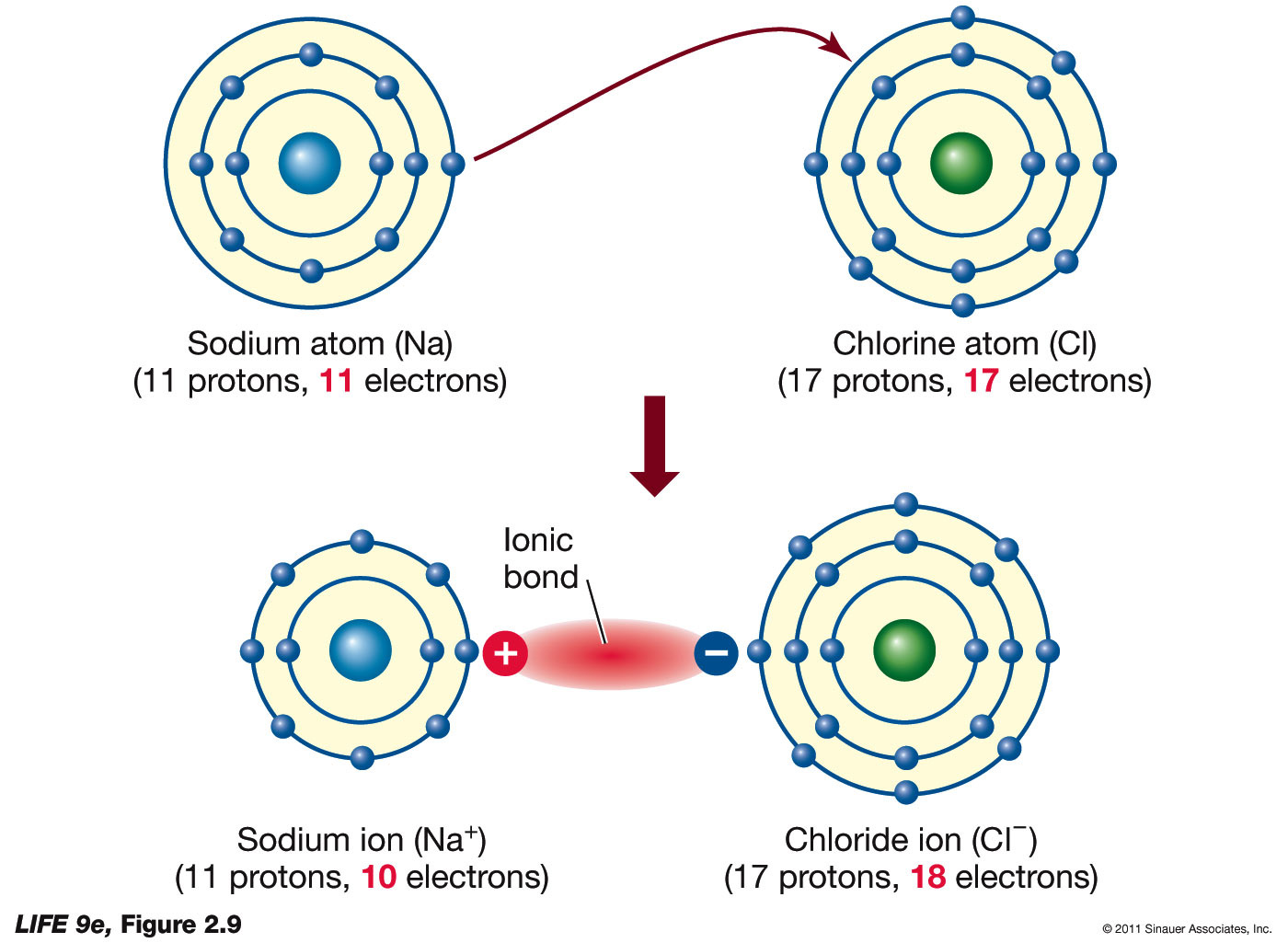
savvy-chemist: Ionic Bonding (2) Dot and cross diagrams/Lewis structures

Ionic Bond Examples | Biology Dictionary

The Ion Propulsion System - What the... #!$@*!! - NaturPhilosophie
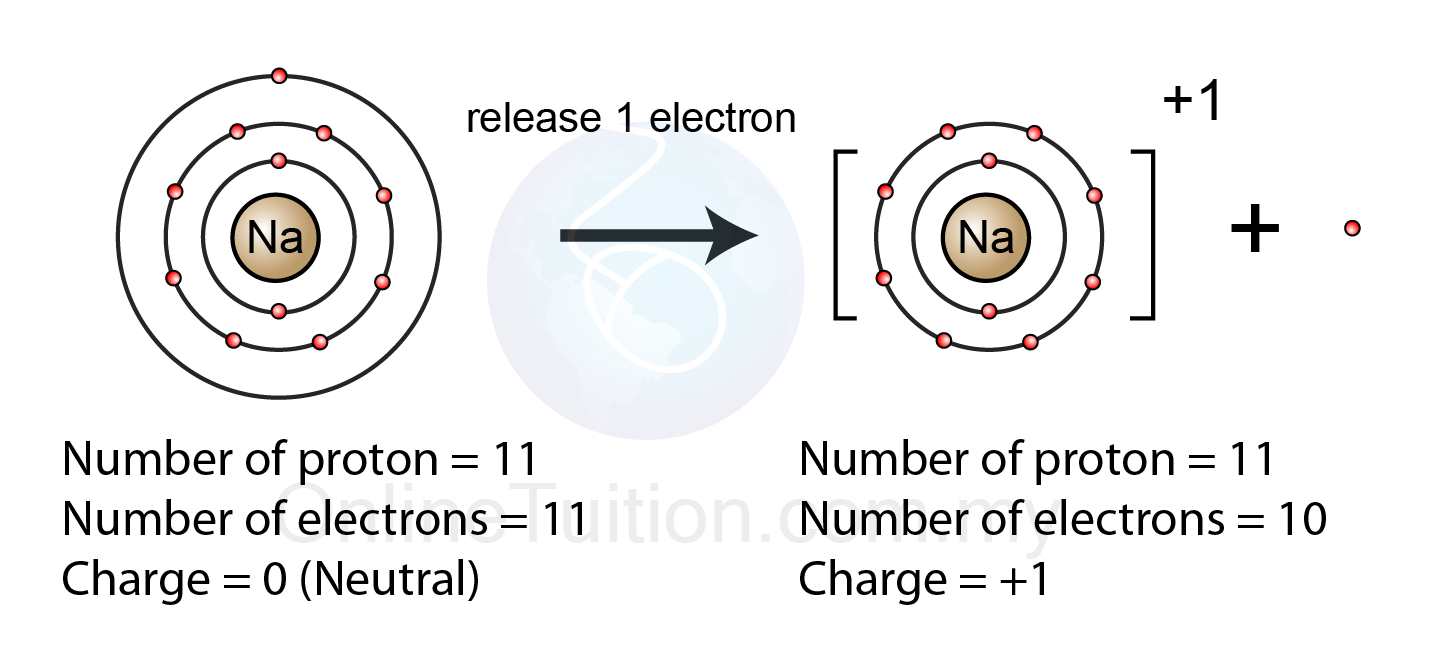
Formation of Ion - SPM Chemistry

Common Polyatomic Ions: Names, Formulae, and Charges | Compound Interest